Silicon Si (Element 14) of Periodic Table Elements FlashCards
sorry for my stutter, i stutter when i talk fast & sorry for any delay that occuredthe website i used is called webwhiteboard.com
Bohr Model Silicon Atom Electron Structure Stock Vector (Royalty Free
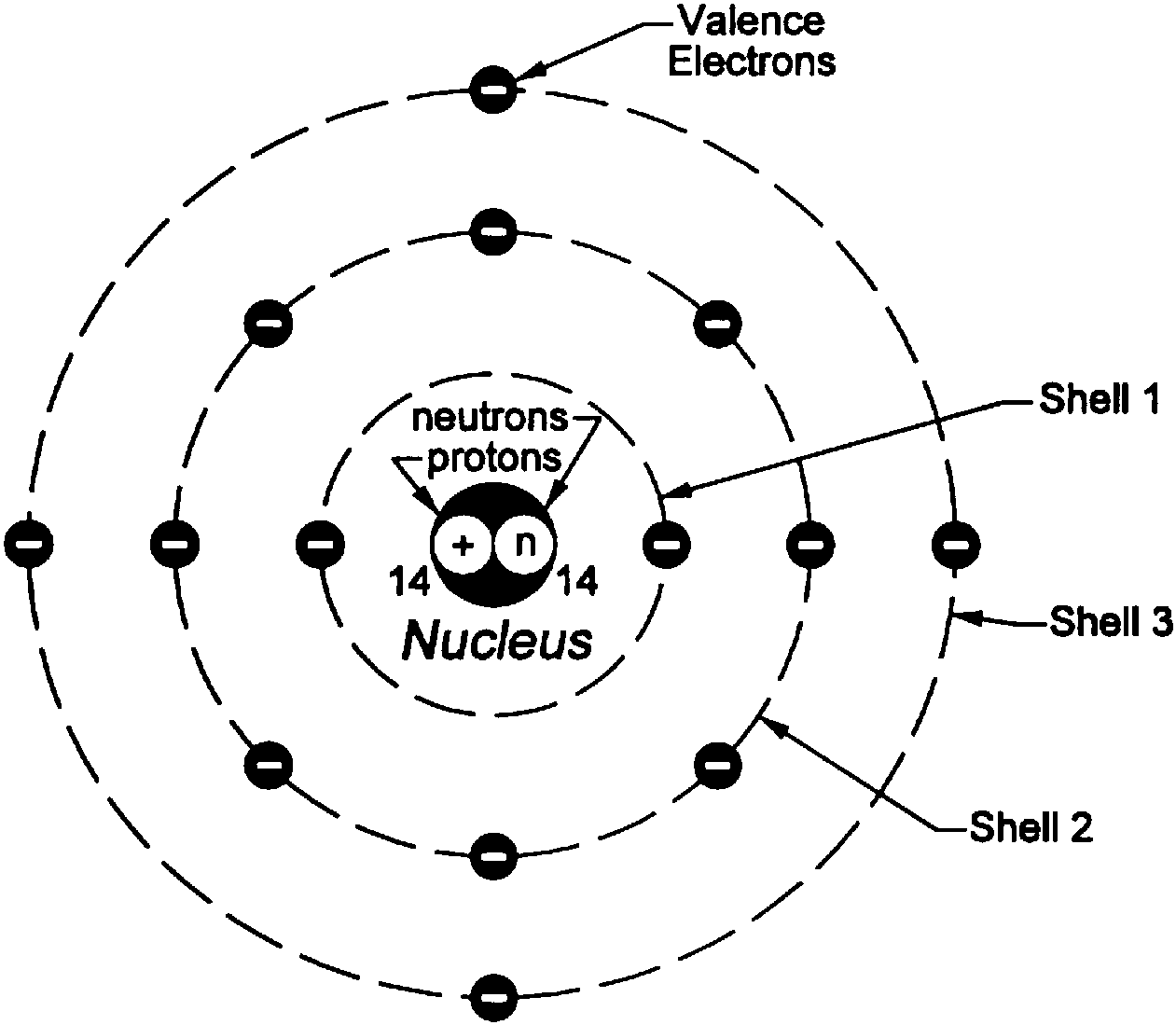
Bohr diagrams show electrons orbiting the nucleus of an atom somewhat like planets orbit around the sun. In the Bohr model, electrons are pictured as traveling in circles at different shells, depending on which element you have. Figure 2 2 contrast the Bohr diagrams for lithium, fluorine and aluminum atoms. The shell closest to the nucleus is.

View 19 Silicon Element Bohr Model aboutdiecolor
Bohr diagram or Bohr rutherford diagram describes the visual representation of orbiting electrons around the small nucleus. It used different electron shells such as K, L, M, N…so on.
Bohr model of silicon atom Electronics And Engineering Lab
Drawing Bohr-Rutherford diagrams is super easy using the following steps: Find the number of protons, neutrons and electrons for the atom. The number of protons is the atomic number. The number of neutrons can be found by subtracting the number of protons from the atomic mass rounded to the nearest whole. This is because protons and neutrons.
Bohr Model For Silicon
In atomic physics, the Bohr model or Rutherford-Bohr model of the atom, presented by Niels Bohr and Ernest Rutherford in 1913, consists of a small, dense nucleus surrounded by orbiting electrons.
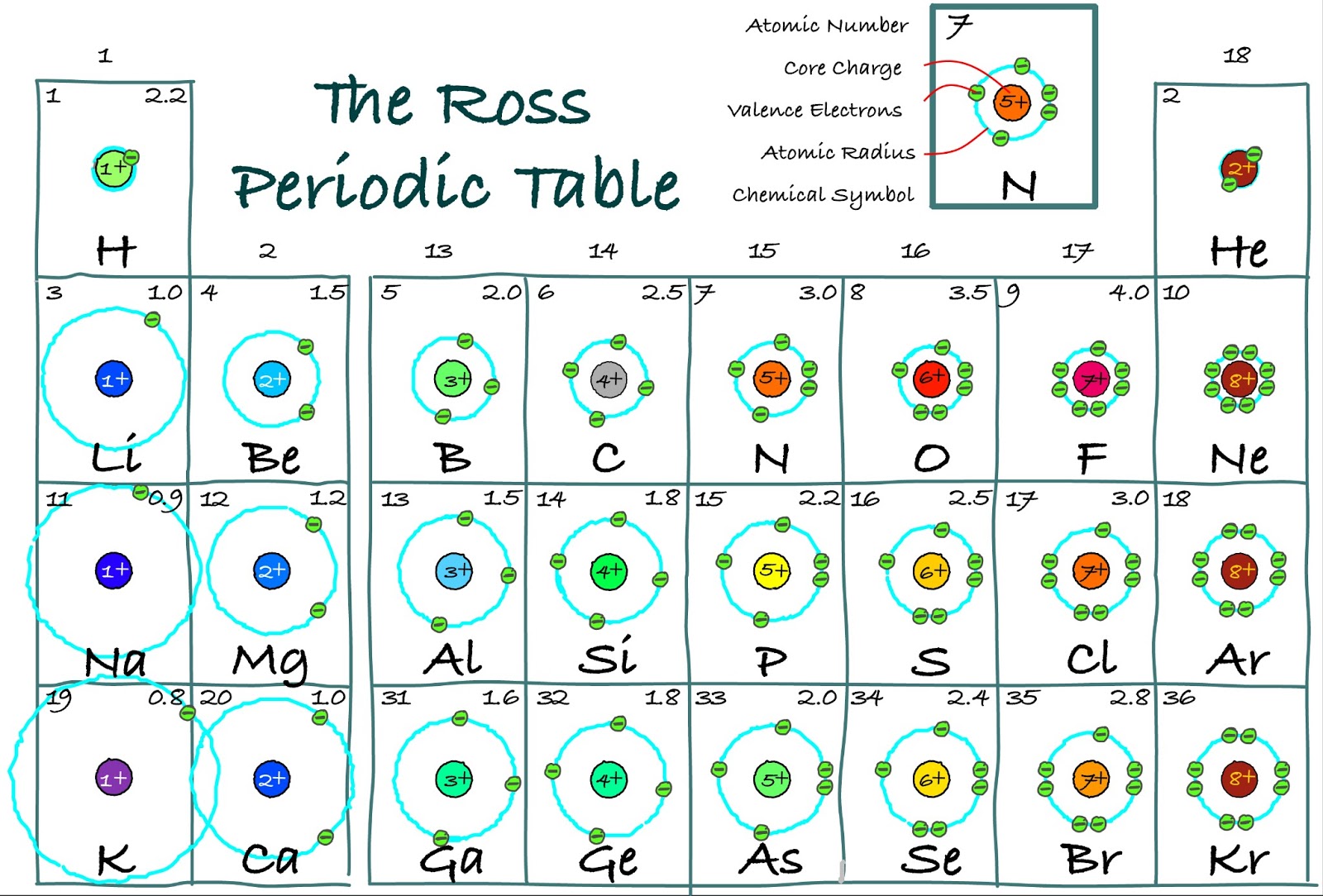
valence electrons of silicon
The Bohr Model has an atom consisting of a small, positively charged nucleus orbited by negatively charged electrons. Here's a closer look at the Bohr Model, which is sometimes called the Rutherford-Bohr Model. Overview of the Bohr Model Niels Bohr proposed the Bohr Model of the Atom in 1915.
Silicon Bohr Model Diagram, Steps To Draw Techiescientist
This page contains materials for the session on the atomic models of Rutherford and Bohr. It features a 1-hour lecture video, and also presents the prerequisites, learning objectives, reading assignment, lecture slides, homework with solutions, and resources for further study.
What Is A Bohr Diagram Hanenhuusholli
The Bohr Model of Silicon (Si) has a nucleus that contains 14 neutrons and 14 protons. This nucleus is surrounded by three-electron shells named K-shell, L-shell, and M-shell. The outermost shell in the Bohr diagram of Silicon contains 4 electrons also called valence electrons. Page Contents show How to draw Bohr Model of Silicon (Si)?
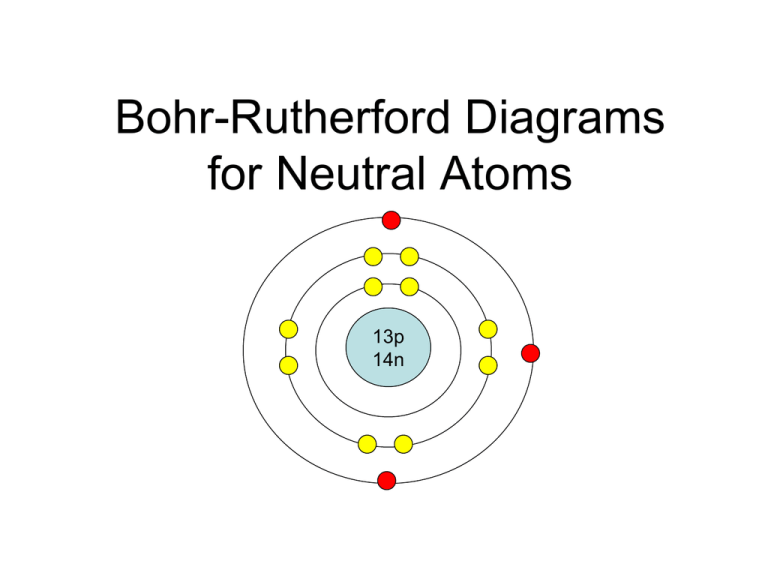
BohrRutherford diagrams for atoms
4) The following diagram is a Bohr-Rutherford diagram of one element from the periodic table: To which group and period does this element belong? A) Period 3 group 4. B) Period 4 group 4. C) Period 3 group 1. D) Period 1 group 3. 5) Referring to the periodic table, find an element that has the same number of electron shells and

Introduction to Electronics Study Guides CircuitBread
draw a Bohr-Rutherford diagram for sulfur. draw a Bohr-Rutherford diagram for chlorine. draw a Bohr-Rutherford diagram for argon. draw a Bohr-Rutherford diagram for potassium. draw a Bohr-Rutherford diagram for calcium. Identification of elements from Bohr-Rutherford Diagram. Learn with flashcards, games, and more — for free.

Introduction to Electronics Study Guides CircuitBread
How to Draw the Bohr-Rutherford Diagram of Silicon chemistNATE 258K subscribers Subscribe Subscribed 245 22K views 4 years ago Silicon has 2 electrons in its first shell, 8 in its second, 4 in.
Symbol and electron diagram for silicon Royalty Free Vector
In this video we'll look at the atomic structure and Bohr model for the Silicon atom (Si). We'll use a Bohr diagram to visually represent where the electrons.
_0.jpg?itok=RuBFwyf1)
Le modèle atomique de RutherfordBohr Alloprof
Mr. Primmer Demonstrates How to Draw Bohr Rutherford Diagrams!
Silicon (Si) AMERICAN ELEMENTS
Manish Bhardwaj. 5.6: Bohr Diagrams of Atoms and Ions is shared under a not declared license and was authored, remixed, and/or curated by LibreTexts. Bohr diagrams show electrons orbiting the nucleus of an atom somewhat like planets orbit around the sun. In the Bohr model, electrons are pictured as traveling in circles at different shells,..

Image result for silicon atomic model
Bohr Diagram: The First Element In order to make a Bohr diagram, you need to know the number of protons, neutrons, and electrons the element has. In this section, we'll show a sample Bohr diagram for hydrogen. H —Hydrogen 1 proton 1 electron 0 neutrons
Electron arrangements
2. What elements is represented by this diagram? How do you know? 3. What is the charge of this nucleus? What is the charge of this atom overall? 4. What is the mass of this atom? 5. Using a periodic table, look up titanium. a. What is its atomic number? b. How many protons does a titanium atom have? c. How many electrons does it have? 6. What.